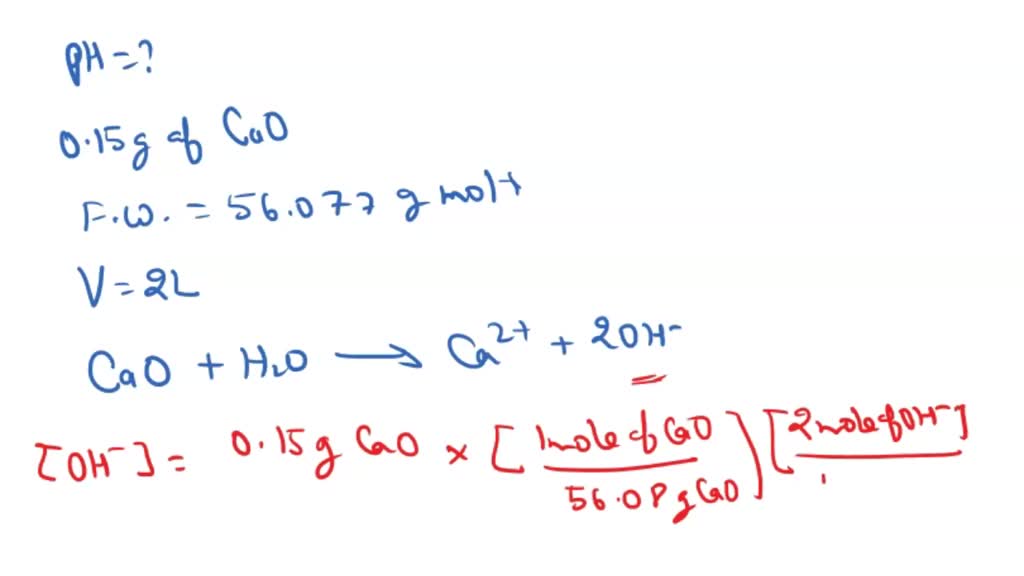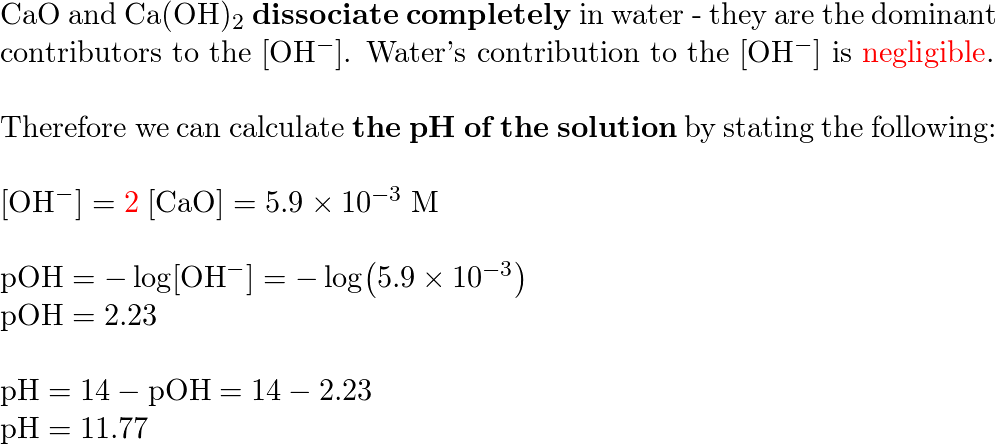
Calcium oxide is a base compound which can react with acids such as nitric acid, HNO3. A student mixed 1.5g of CaO with 2.5M HNO3. The chemical equation for this reaction is:

SciELO - Brasil - Strawberry (Fragaria X ananassa Duch.) yiel das affected by the soil pH Strawberry (Fragaria X ananassa Duch.) yiel das affected by the soil pH

SOLVED: Question 34 (1 point) CaO, lime, is used in agriculture to raise the pH of soil How does this happen? CaO forms CaCO3 in the soil None of the choices CaO

A solution is prepared by dissoving 2.8 g of lime (CaO) in enough water to make 1.00 L of lime - YouTube

pH change of oxygen controlled-release microspheres (OCRMs) and CaO 2... | Download Scientific Diagram