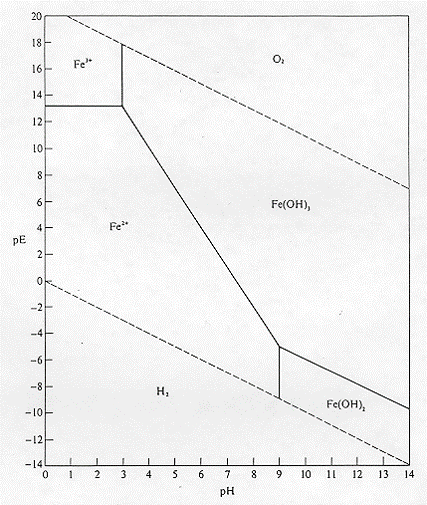
![PDF] The effect of pH on the kinetics of spontaneous Fe(II) oxidation by O2 in aqueous solution--basic principles and a simple heuristic description. | Semantic Scholar PDF] The effect of pH on the kinetics of spontaneous Fe(II) oxidation by O2 in aqueous solution--basic principles and a simple heuristic description. | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/f83dd855e637c2cd9ad1f9e0b2255706cb9d29e3/2-Figure3-1.png)
PDF] The effect of pH on the kinetics of spontaneous Fe(II) oxidation by O2 in aqueous solution--basic principles and a simple heuristic description. | Semantic Scholar

The effect of pH on the kinetics of spontaneous Fe(II) oxidation by O2 in aqueous solution – basic principles and a simple heuristic description - ScienceDirect
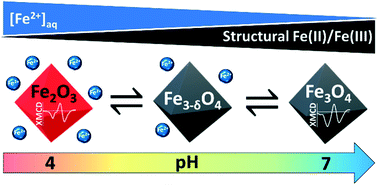
Prediction of nanomagnetite stoichiometry (Fe(ii)/Fe(iii)) under contrasting pH and redox conditions - Environmental Science: Nano (RSC Publishing)

The effect of pH on the kinetics of spontaneous Fe(II) oxidation by O2 in aqueous solution – basic principles and a simple heuristic description - ScienceDirect

Speciation of 0.5 mM Fe(III) between pH 1 and 4 at 25 ○ C and an ionic... | Download Scientific Diagram

Solubility of Fe(III) vs pH, as controlled by the solubility of 2-line... | Download Scientific Diagram

A pe-pH diagram of Fe speciation in soil at 25°C, partial pressures of... | Download Scientific Diagram

Toplam toprak analizi (Fe,Cu,Zn,Mn,Ca,Mg,pH,tuz,kireç,bünye - İŞBA,N,P,K) - ürününü globalpiyasa.com da satın alın







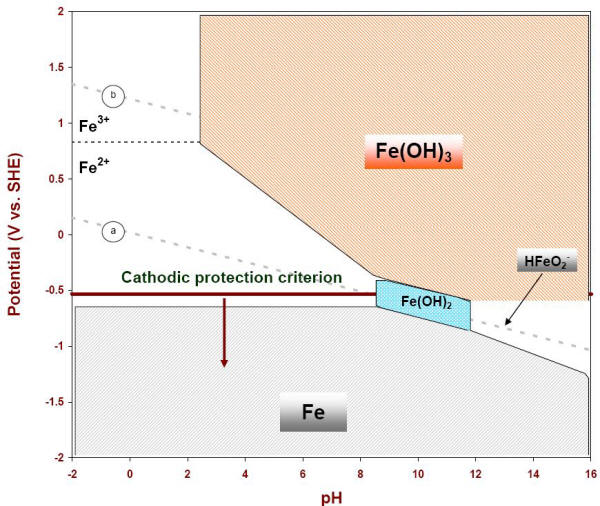

![The speciation of Fe(III) in seawater as a function of pH 25 °C.[5] | Download Scientific Diagram The speciation of Fe(III) in seawater as a function of pH 25 °C.[5] | Download Scientific Diagram](https://www.researchgate.net/publication/26429899/figure/fig9/AS:281764753887234@1444189360252/The-speciation-of-FeIII-in-seawater-as-a-function-of-pH-25-C5.png)