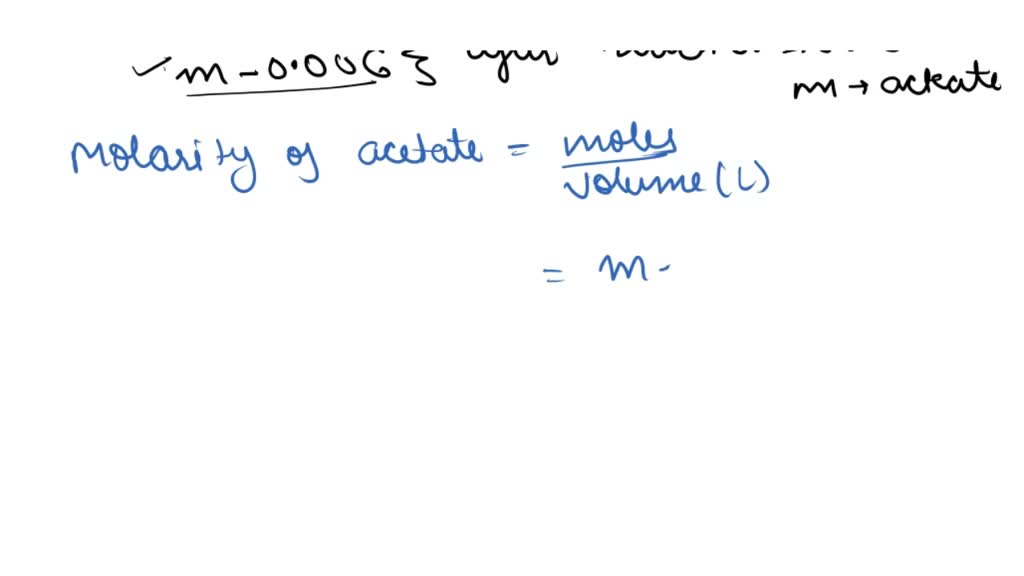
SOLVED: 1.0 mL of 6.0 M HCl is added to 100 mL of a buffer that contains equal moles of acetic acid (CH3COOH, Ka = 1.8 × 10-5) and sodium acetate (Na (CH3COO)).
Why does the solution of sodium acetate give more concentration of Hydroxide ion? Shouldn't the number of Hydroxide ion and hydrogen ion be equal? - Quora
/is2.ecplaza.com/ecplaza2/products/8/8f/8f7/1187082394/sodium-sodium-ethanoate.jpg)
Sodium sodium ethanoate Acetate CH3COO Na NaOAc C2H3NaO2 C2H3NaO2-3H2O - Lianyungang Zhaofu Mineral Co.,Ltd.

Calculate the mass of sodium acetate (CH3COONa) required to make 500 ml of 0.375 molar aqueous solution. Molar mass of sodium acetate is 82.0245 g mol^-1 .
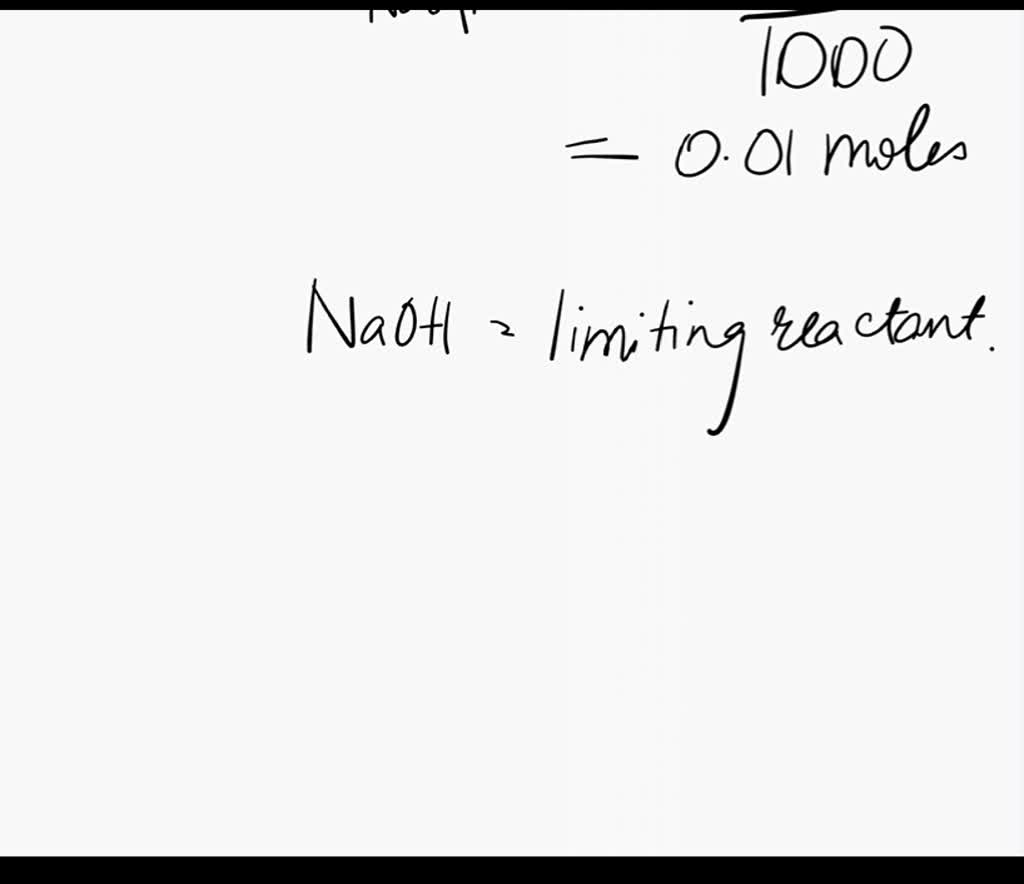
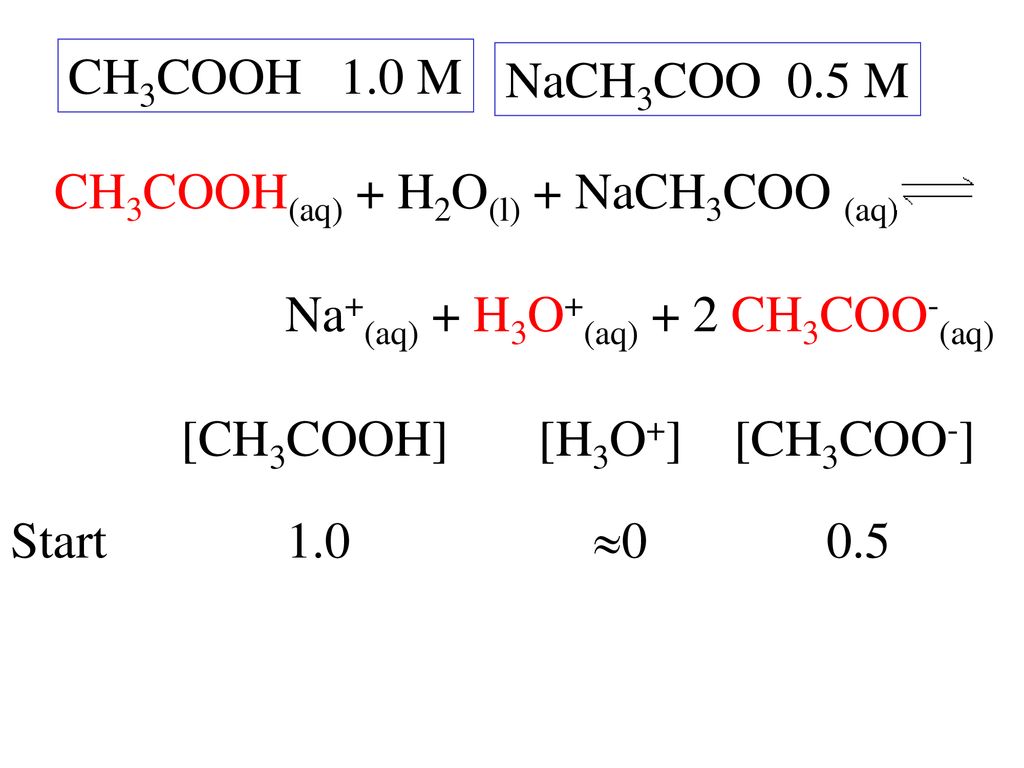
SOLVED: If 10 mL of 1M NaOH are added to one liter of a buffer that is 0.3 M acetic acid and 0.2 M sodium acetate (Na+CH3COO–), how much does the pH

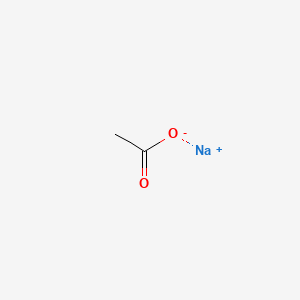
Sodium Acetate(CH3COONa) - Structure, Properties, Preparations, Uses, Important questions, FAQs of sodium acetate.

What is the pH of a 0.402 M aqueous solution of NaCH3COO? Ka (CH3COOH) = 1.8x10-5 | Homework.Study.com