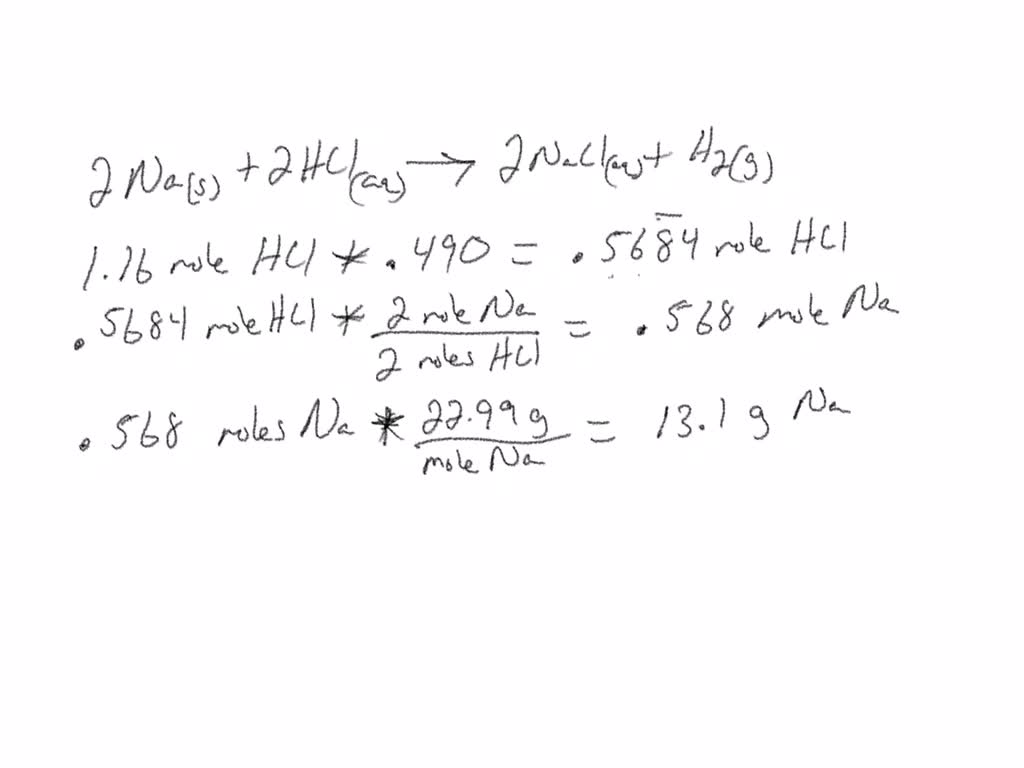Na+HCl=NaCl+H2 balance the chemical equation @mydocumentary838. na+hcl=nacl+h2 balance the equation - YouTube

Hydrochloric Acid, c(HCl)=0.1 mol/L (0.1N) Titripur , MilliporeSigma, Quantity: 1 L | Fisher Scientific

How to balance Na+HCl=NaCl+H2|chemical equation Na+HCl=NaCl+H2|Na+HCl=NaCl+H2 balance equation - YouTube
Fluka 35334 Hydrochloric Acid Solution Volumetric, 0.01 M Hcl (0.01N) Analiz grade Plastic Bottle 1 L

sodium reacts with Hydrogen chloride and gives rise sodium chloride and hydrogen .Balance the - Brainly.in
110ml of N/2 HCl, 20 ml of M/5 H2SO4,20 ml of N/4 H3PO4 are mixed together.The volume of water that must be added to make a 0.18N solution is(assume there is no

Ca , Na , Al , Cu , Pt , ile HCl ve NaOH tepkimesi ne oluır sırasıyla tepkimesi ne olur hepsi sırasıyla - Eodev.com

SOLVED: How many mL of 0.250 M HCl would react exactly with 30.0 mL of the 0.150 M solution of Na(OH)2 solution? The chemical reaction involved is: HCl(aq) + NaOH(aq) → NaCl (