
SOLVED: Calculate the solubility of silver chromate, Ag2CrO4, in 0.005 M Na2CrO4. Ksp = 2.6 ´10-12 1) 1.4 ´ 10-4 M 2) 1.6 ´ 10-6 M 3) 3.4 ´ 10-5 M 4) < 1.0 ´ 10-6 M 5) 1.1 ´ 10-5 M

Jual Na2CrO4, Natrium kromat, Sodium Chromate, AR - 10gr - Kab. Bandung Barat - Arsyil Pradipta Chemical | Tokopedia

310 g of a carbohydrate leaves a residue of 124 g of carbon on heating in absence of air. If the empirical formula of the carbohydrate is CxHyOz , then find the

Sodium chromate (Na2CrO4) solution in a test tube. The yellow colour comes from the chromium ions rather than the sodium ions Stock Photo - Alamy




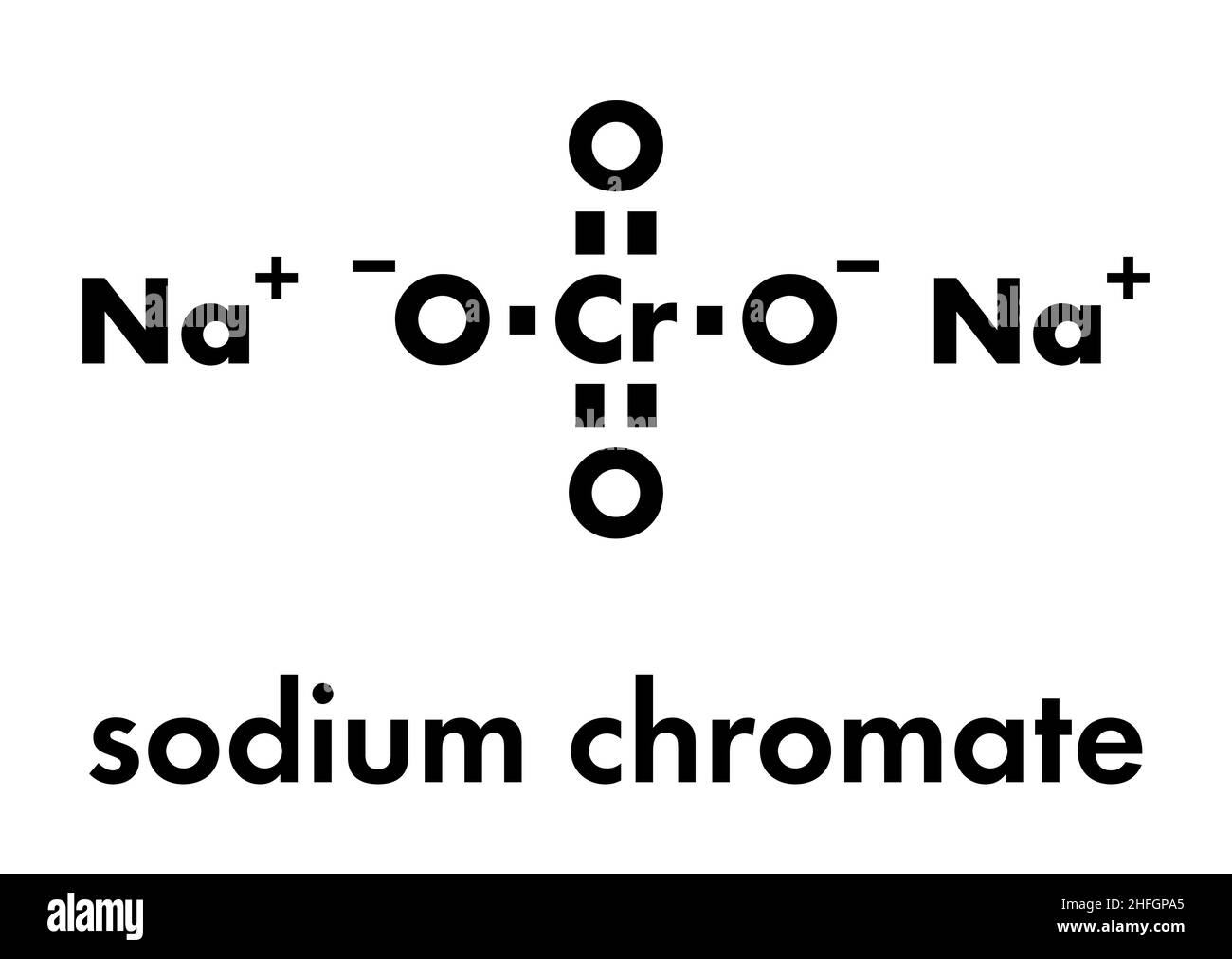

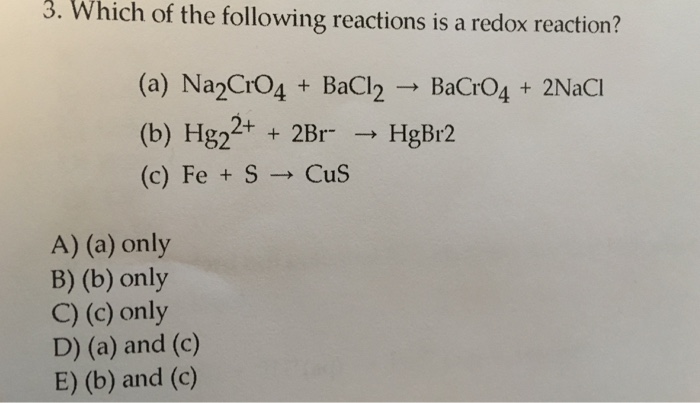



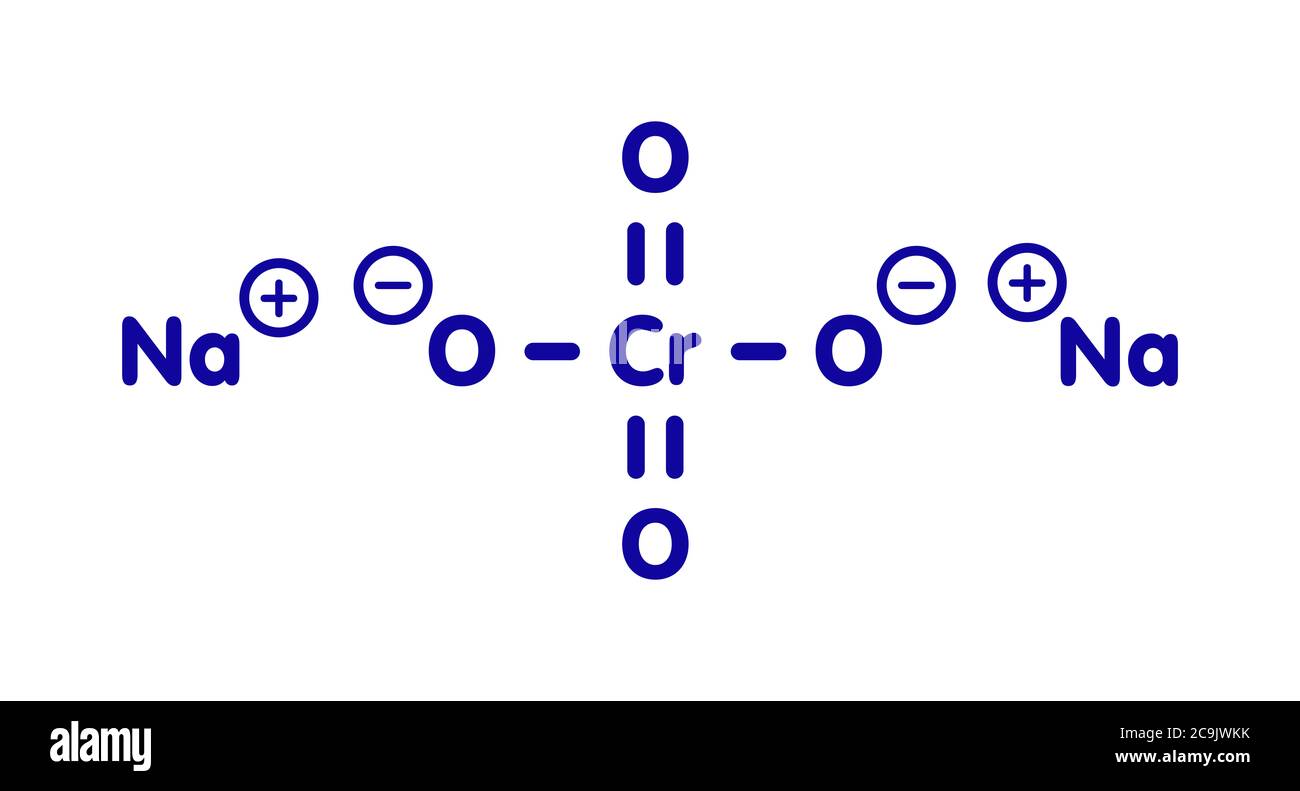







![ANSWERED] Consider the balanced reaction below: Pb(... - Physical Chemistry ANSWERED] Consider the balanced reaction below: Pb(... - Physical Chemistry](https://media.kunduz.com/media/sug-question/raw/70546283-1660059482.6685135.jpeg)

