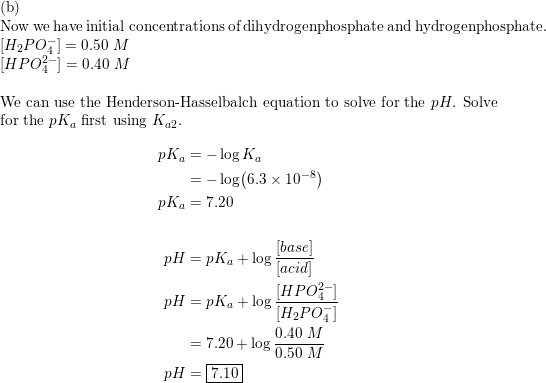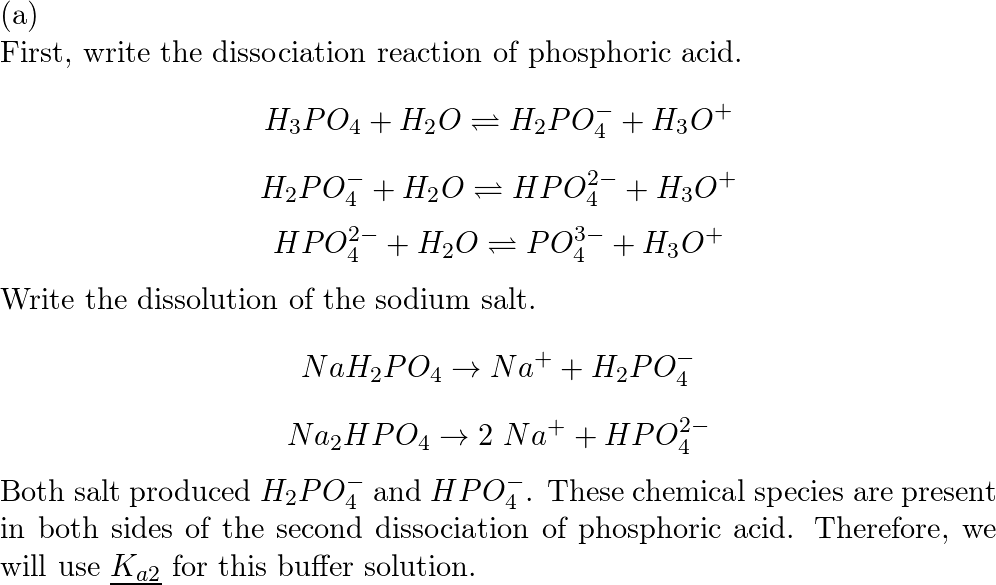Calculate pH a) NaH(2)PO(4) b) Na(2)HPO(4) respectively, for H(3)PO(4) pKa(1) = 2.25, pKa(2) = 7.20, pKa(3) = 12.37)

Calculate pH of a 0.1 M Na2HPO4 solution. K1, K2 and K3 for H3PO4 are 7.1 × 10^-3 × 6.3 × 10^-8 and 4.5 × 10^-13 respectively. Which approximation is necessary for the calculation?

SOLVED: 1. What would be the pH of a solution prepared by combining equal quantities of NaH2PO4 and Na2HPO4? Explain.2. Sufficient strong acid is added to a solution containing Na2HPO4 to neutralize

Table 2 from Dynamic approach to predict pH profiles of biologically relevant buffers | Semantic Scholar

SWV of FEN (5.0x10-5 mol L-1 ) in Na2HPO4 0.1 mol L-1 , pH 4.0, f= 100... | Download Scientific Diagram
Potential response characteristics in 0.1 M Na2HPO4 solution at pH 9.0... | Download Scientific Diagram

Table 1 from Dynamic approach to predict pH profiles of biologically relevant buffers | Semantic Scholar