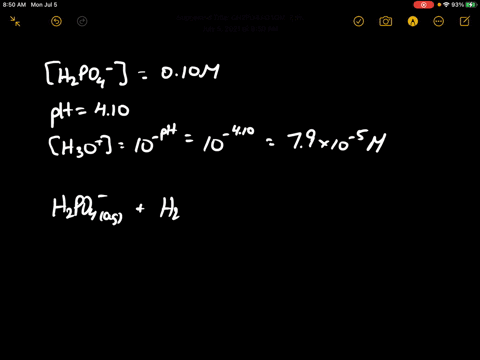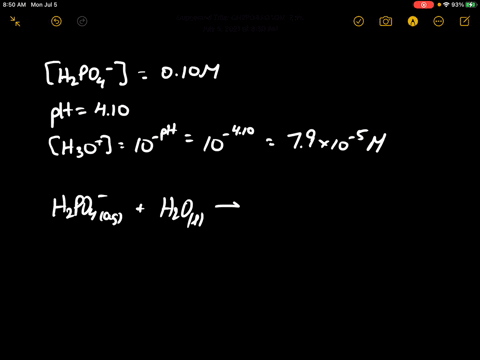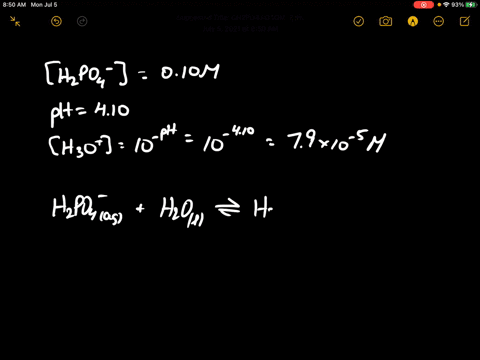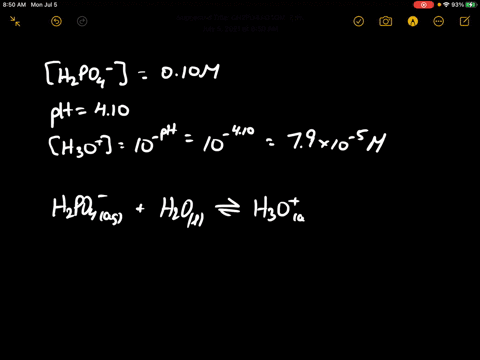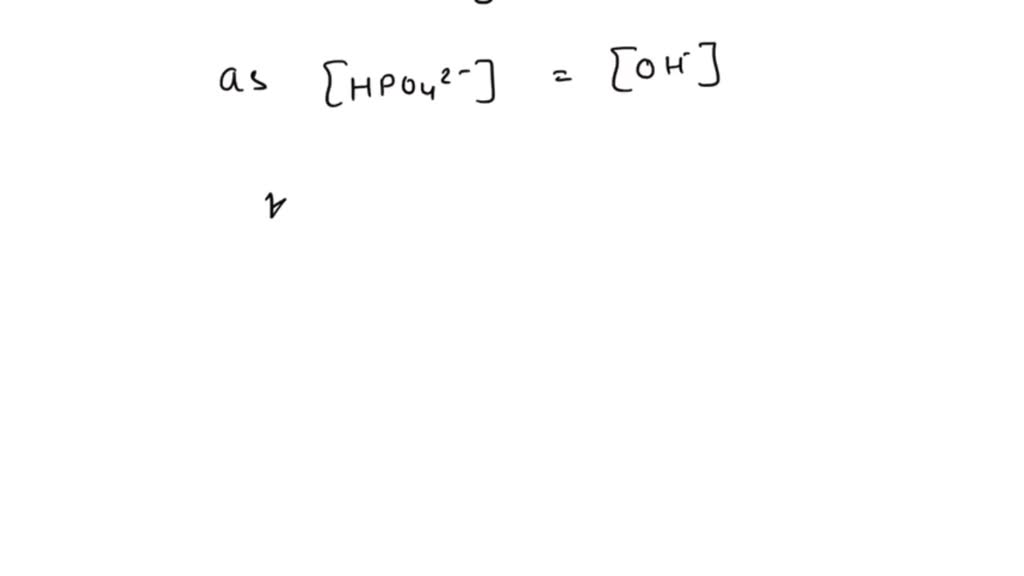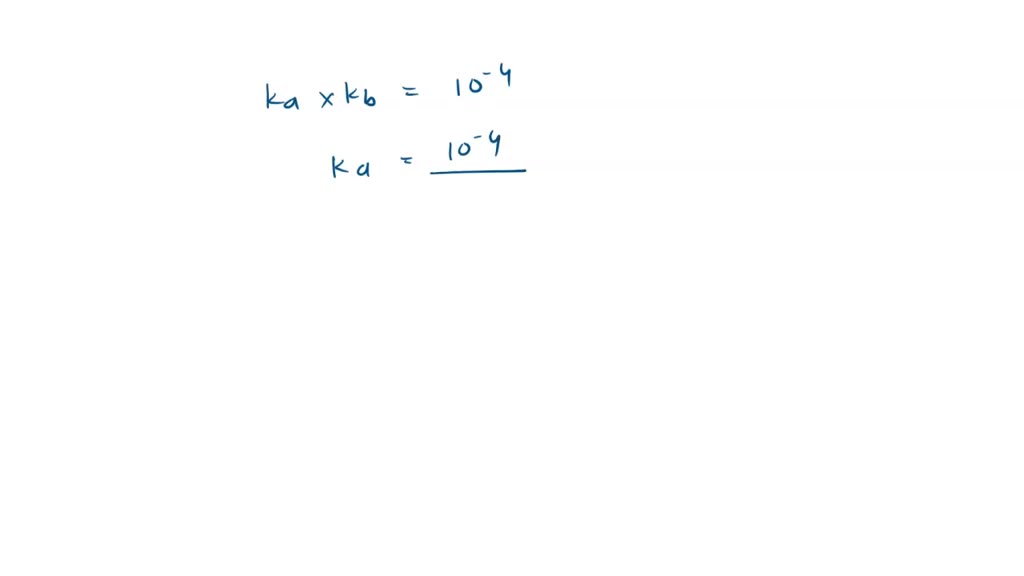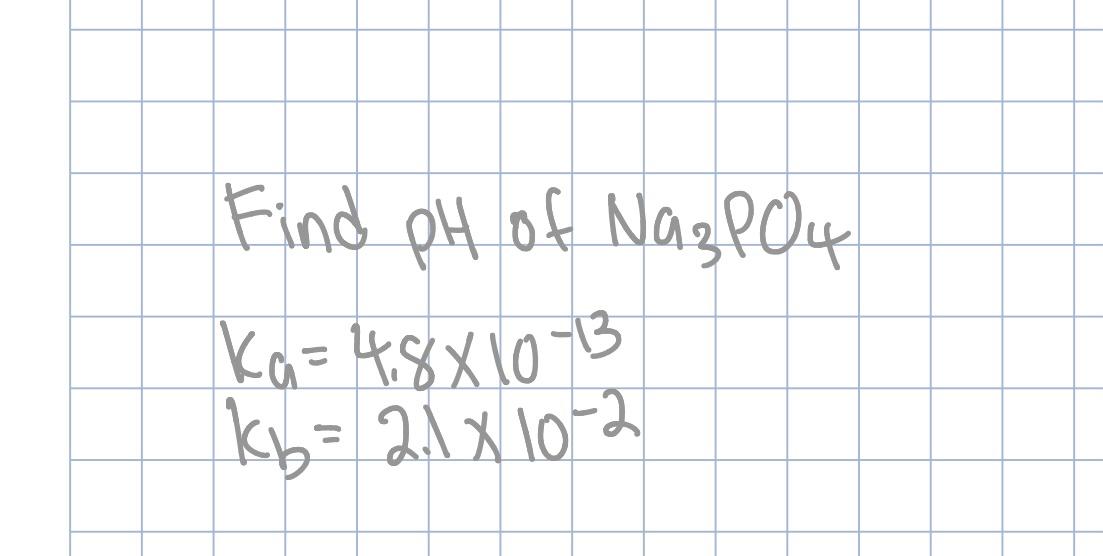When 100 mL of 0.1 M KNO3 , 400 mL of 0.2 M HCl and 500 mL of 0.3 M H2SO4 are mixed, then in the resulting solution :
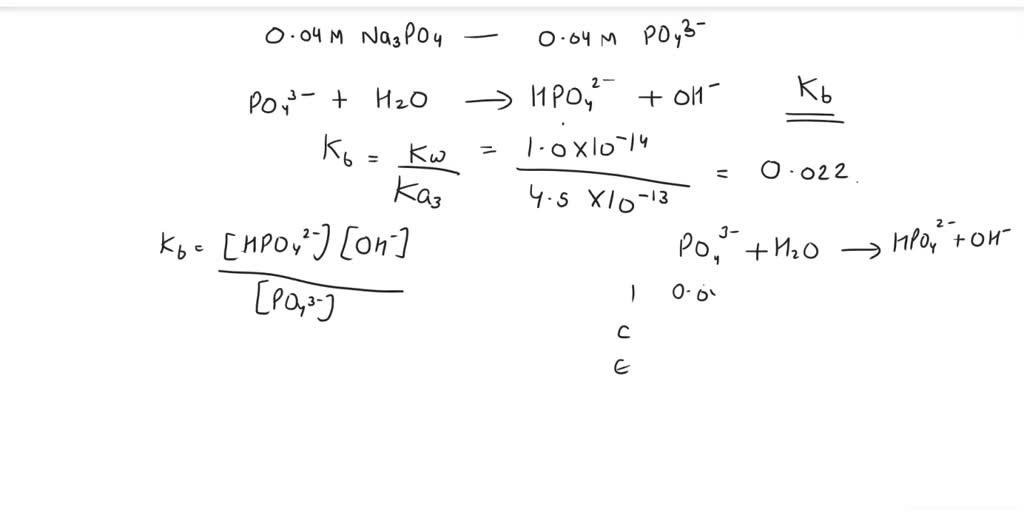
SOLVED: Find the pH of the solution containing 0.04 M Na3PO4. Ka1 = 7x10-3 , Ka2= 2.2x10-7 , Ka3= 4. 5x10-13 can you please explain it step by step?

When 100 mL of 0.1 M KNO3 , 400 mL of 0.2 M HCl and 500 mL of 0.3 M H2SO4 are mixed, then in the resulting solution :
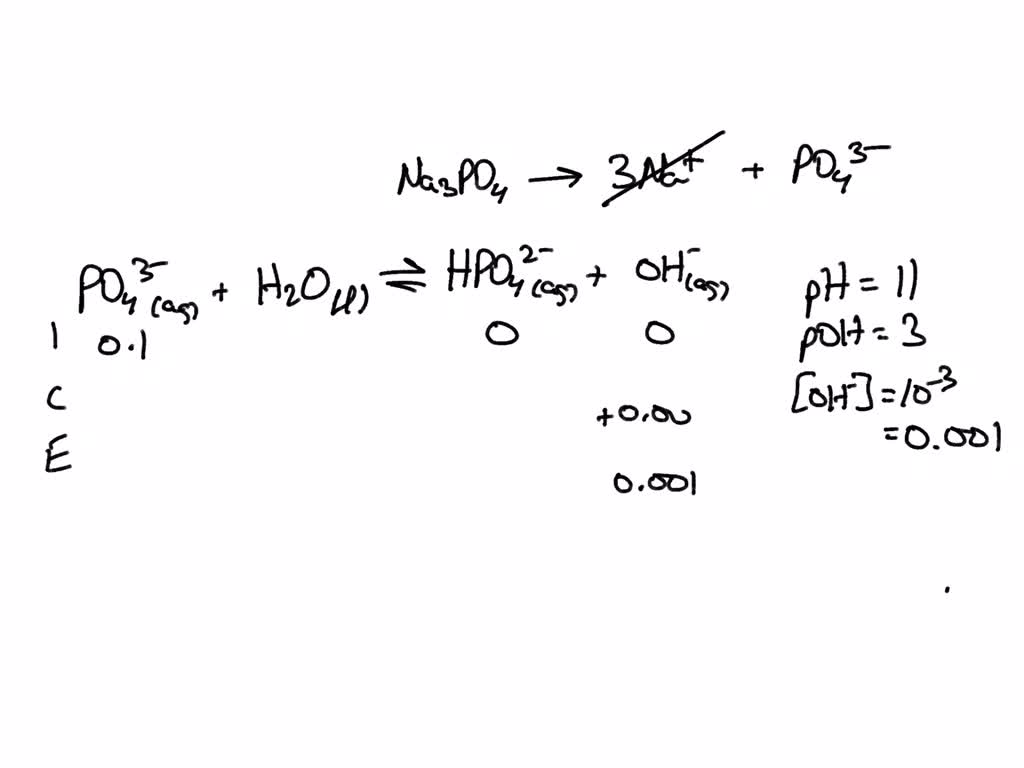
Calculate the pH of 0.5 M Na3PO4 in aqueous solution ? - Sarthaks eConnect | Largest Online Education Community
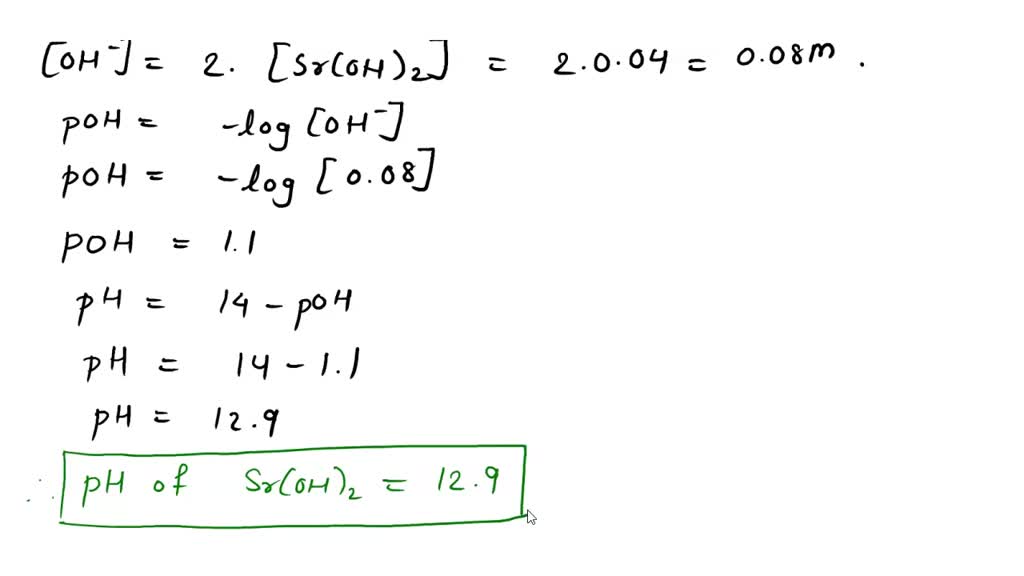
SOLVED: 2.Calculate the pH of the following solutions a.0.125M H3BO3 b.0.200 M Na2CO3 c.0.0746 M H2S d.0.225 M Na3PO4
What is the pH of 1.0 M Na3PO4 in aqueous solution ? - Sarthaks eConnect | Largest Online Education Community