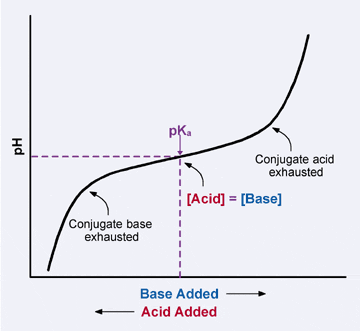
The rapid change of pH near the stoichiometric point of an acid - base titration is the basis of indicator detection. The pH of the solution is related to the ratio of

Finding the pKa Values of a Double-Range Indicator Thymol Blue in a Remote Learning Activity | Journal of Chemical Education
the pKa of HNO_2 IS 3.37.The pH of HNO_2 in its 0.01 mol/l aqueous solution will be 1.5.37 2.2.69 3.1.69 4.0.69

PH maddeler tampon tozu PH ölçer tedbir kalibrasyon çözeltisi PH 4.00 potasyum hidrojen ftalat 10 / PK|powder powder|powder lotpowder measure - AliExpress

How should the acid dissociation constant pKa be measured? | Automatic Potentiometric Titrators | Faq | Kyoto Electronics Manufacturing Co.,Ltd.("KEM")












:max_bytes(150000):strip_icc()/what-is-pka-in-chemistry-605521_FINAL2-9fdfc39e9aa34caa96d6e74a2c687707.png)
![Solved Just as pH is the negative logarithm of (H30+], pK, | Chegg.com Solved Just as pH is the negative logarithm of (H30+], pK, | Chegg.com](https://media.cheggcdn.com/media/375/3755951c-9b2e-4931-a914-33bb100113d8/php72x3Cn.png)
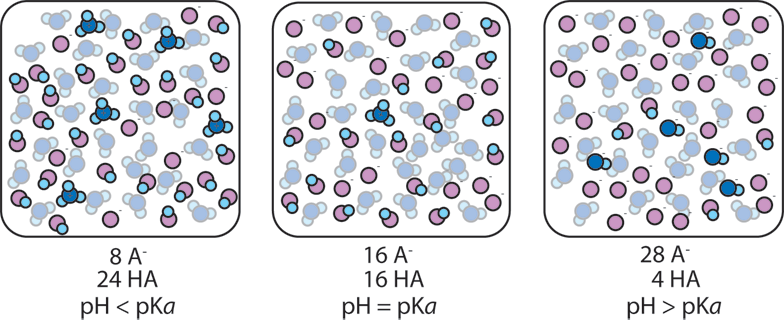
![SOLVED: [A-] log [HA] pKa pH d. [A- ] PH = PKa + log [HA] [A-] PH = pKa + [HA] SOLVED: [A-] log [HA] pKa pH d. [A- ] PH = PKa + log [HA] [A-] PH = pKa + [HA]](https://cdn.numerade.com/ask_images/959424b8320b49c898de609f834a314d.jpg)